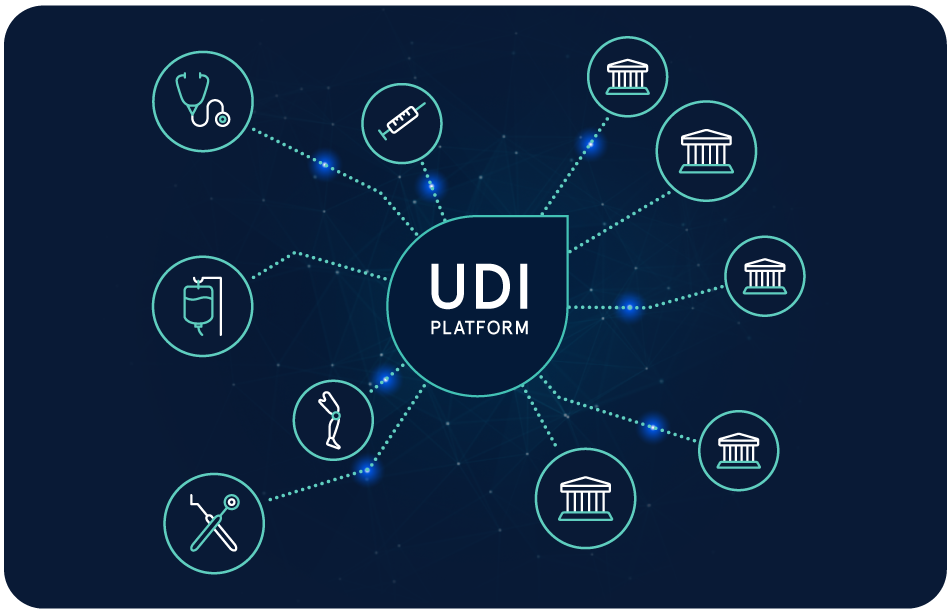
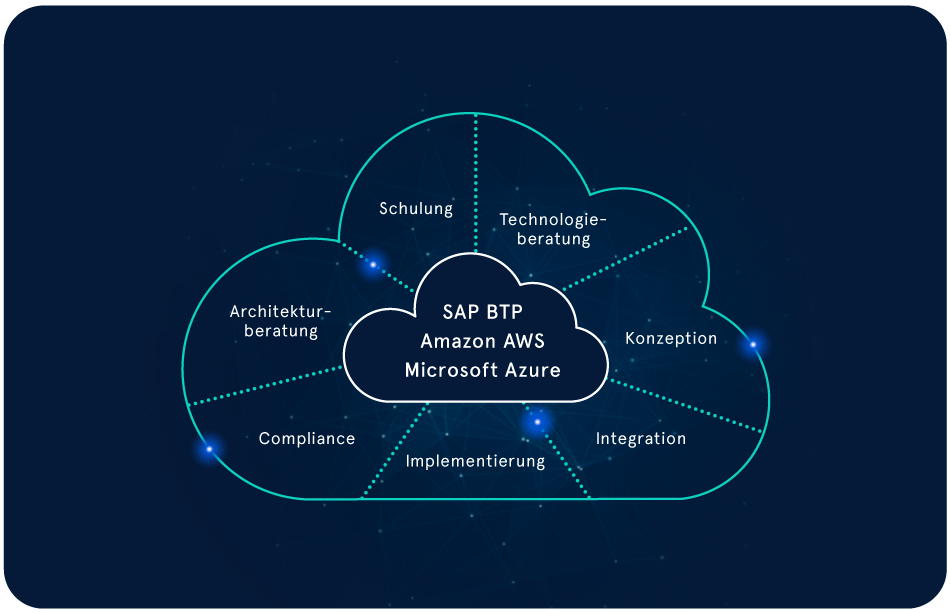
Die Zukunft von Life Sciences gestalten – mit innovativen Cloud-Lösungen
Wir helfen Life Sciences-Unternehmen, Prozesse zu optimieren, Kosten zu reduzieren und die Zusammenarbeit zu vereinfachen. Mit unseren Cloud-Lösungen können Sie sich auf Ihre Kernkompetenzen fokussieren, Innovationen schneller auf den Markt bringen und das Leben von Patienten verbessern.